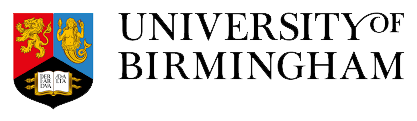Awarded Projects
This page highlights the projects supported through our funding schemes, including fellowships, flexible funds, mobility awards, and engagement and communication awards. Together, these opportunities enable innovative research, foster interdisciplinary collaboration, and strengthen connections across the wider community.
First wave of GIBA flexible funding awards revealed
The UK GIBA Network+ has announced the first projects to receive funding through its Flexible Funds scheme, supporting innovative interdisciplinary research into the gut–immune–brain axis.
The inaugural awards span studies exploring how gut barrier integrity links to neurobehavioural outcomes, whether nutritional interventions can boost nervous system function after menopause, how ageing reshapes neuroimmune interactions in the gut, and whether probiotics can improve wellbeing by reducing inflammation. Together, these projects will pump-prime collaborative research that deepens mechanistic understanding, integrate novel technologies and help forge long-term partnerships across the UK research community.
Read on for more details of the four awarded projects:
Systemic integration of gut microbial modulation effects across the Gut-Immune-Brain Axis (GIBA)
- Lead applicant: Andrea Monteagudo-Mera, University of Reading
- Co-applicant: Antonio Riva, Foundation for Liver Research & King’s College London
This project will investigate how gut barrier integrity and immune variability relate to other components of the gut-immune-brain axis. The team will generate new systemic immunological profiles to complement and integrate neurobehavioural and cognitive (resting-state fMRI, magnetic resonance spectroscopy, behavioural measures including facial emotion recognition, motor performance and tactile sensory function), microbiome, and metabolomics data from an existing large cohort of healthy individuals. This dataset also includes subsets of participants exposed to gut microbial modulation interventions (probiotics).
This ‘systems biology’ approach is essential to gain deeper insights into the integrated GIBA mechanisms across the body, and how gut, immune and neurobehavioural processes link together.
Alpha-lactalbumin supplementation to counter serotonergic gut-brain-axis impairment after the menopause
- Lead applicant: Paul Ansdell, Northumbria University
- Co-applicant: Kirsty Hicks, Northumbria University

This project will test whether a nutritional supplement (alpha-lactalbumin, a protein powder rich in the amino acid tryptophan) can enhance nervous system function in post-menopausal women.
Researchers have previously demonstrated that the spinal cord loses excitability following the menopause, a key physiological quality underpinned by the neurotransmitter serotonin. This excitability is critical for maintaining our ability to produce force and stay independent as we move from midlife to older age. Hormone therapy can counter this decline, but not all women are eligible for or wish to use it, so non-pharmacological alternatives must be explored.
Most of the body’s serotonin is produced in the gut, meaning what we consume can directly influence brain chemistry and nervous system function. In healthy young adults, this supplement has been shown to increase serotonin levels, but it has never been tested in post-menopausal women. This project will therefore investigate whether the supplement can increase serotonin levels, enhance nervous system excitability, and improve strength and coordination in this population.
Spatial transcriptomics to examine the effect of aging on neuroimmune interactions in the human intestine – a pilot study
- Lead applicant: Michael FitzPatrick, University of Oxford
- Co-applicant: Agne Antanaviciute, University of Oxford
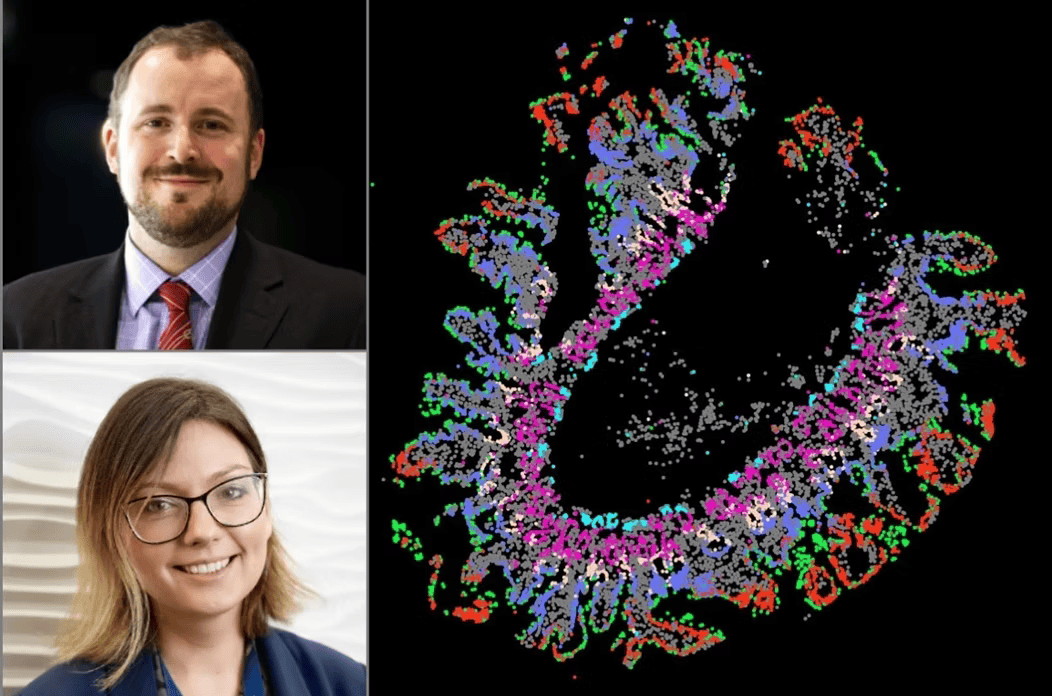
As we age, the gut’s nervous system (called the enteric nervous system, or ENS) starts to decline.
For example, certain immune cells that normally help protect nerves switch to a more inflammatory state with age, harming nerve health. Hormone-releasing cells also become less active, reducing signals that keep gut movement normal. Changes in gut bacteria make things worse by reducing beneficial compounds that support nerve health.
To study these complex interactions, researchers are using advanced techniques that allow them to look at cells in their natural positions within tissue, rather than isolating them. This includes spatial transcriptomics, which maps gene activity in tissue slices, combined with special staining to visualise nerve structures. The goal is to create detailed maps of how nerves, immune cells, and other gut cells interact—and how these relationships change with age.
This research will start with a pilot study comparing gut tissue from younger and older people. The findings will help build a bigger project aimed at understanding how aging affects gut health and could eventually lead to new ways to keep our digestive system healthy as we grow older.
A randomised controlled trial investigating the probiotic-mediated suppression of inflammatory cytokines as a mechanism for improving wellbeing
- Lead applicant: Patricia Sanz Morales, University of Reading
- Co-applicant: Prof Philip Burnet, University of Oxford
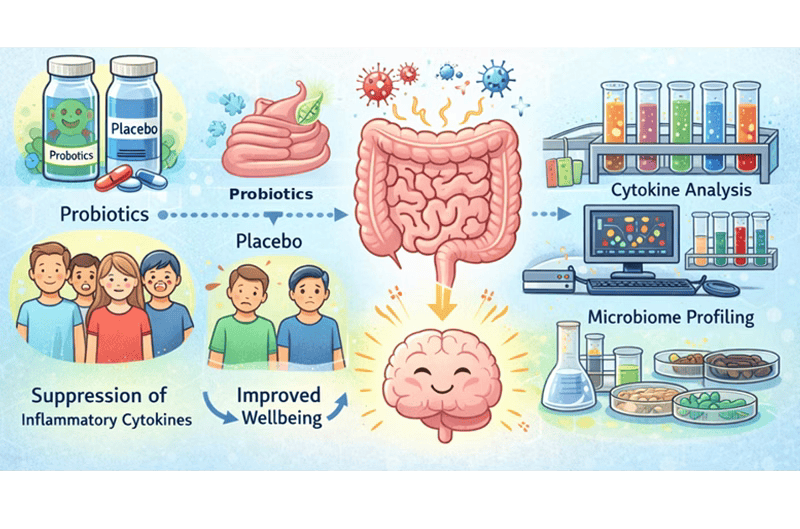
This project will test whether lowering circulating inflammatory mediators can improve emotional wellbeing and everyday cognitive function.
This study will evaluate the effects of a multi-strain probiotic on circulating cytokines and wellbeing through a randomised double-blind, placebo-controlled trial. The intervention is a probiotic selected as a safe, acceptable and scalable mechanistic probe that perturbs the gut environment without adverse clinical effects.
Researchers will use a UK-licensed, quality-controlled, preparation with a matched placebo as a standardised, well-documented tool. The primary target is pre-specified as a baseline-adjusted pro-inflammatory composite, chosen for sensitivity and reproducibility in nutrition/microbiome trials and because these axes show the most consistent links to mood and prospective risk.
Pre-registered mediation analyses will test whether immune change explains part of any improvement in wellbeing. This integrated approach strengthens inference, delivers effect sizes and standardised methods, and provides openly shared resources to de-risk a larger multicentre RCT aligned with GIBA’s mechanistic remit.